
Federal health officials have issued a nationwide recall for two moringa‑based dietary supplements after linking them to a multistate Salmonella Typhimurium and Salmonella Newport outbreak that sickened 97 people across 32 states. Twenty‑six individuals were hospitalized; no deaths were reported.



Recalled Products
• Why Not Natural Pure Organic Moringa Green Superfood capsules
• Lot: A25G051
• Expiration: 07/2028
• Live it Up Super Greens dietary supplement powder
• Flavors: Original, Wild Berry
• Lots: All beginning with “A”
• Stick packs: Expiration 08/2026–01/2028
Products were sold online, including company websites, Amazon, eBay, and Walmart.
Outbreak Findings
Testing by FDA and state partners found outbreak strains of Salmonella in six samples, including:
• Contaminated moringa leaf powder ingredients
• Opened Why Not Natural capsules
• Opened and unopened Live it Up Super Greens powders
The FDA has closed its investigation, but warns that these supplements have long shelf lives, and contaminated product may still be in homes.
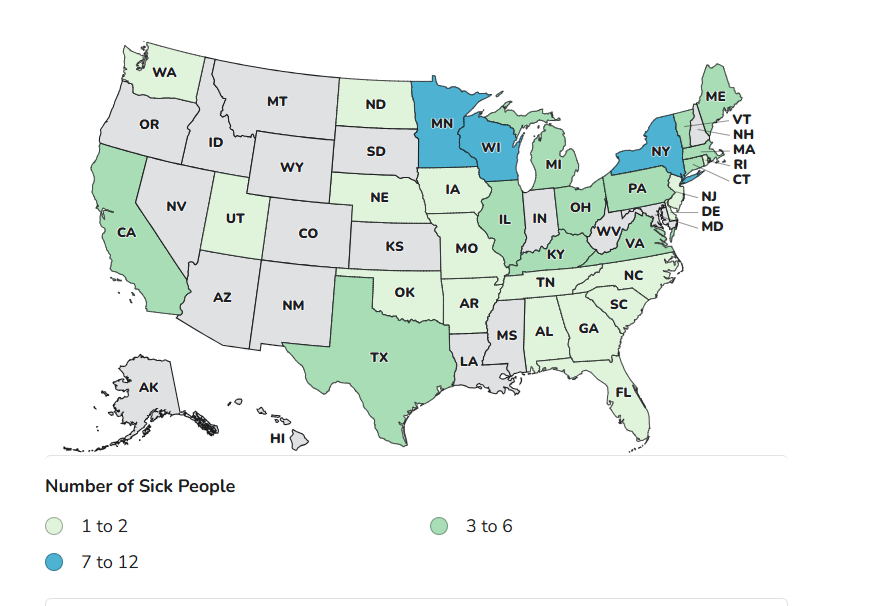 Illness Summary
Illness Summary
• Illnesses: 97
• Hospitalizations: 26
• Deaths: 0
• Last illness onset: February 27, 2026
• Distribution: Nationwide, including Puerto Rico, Guam, and the U.S. Virgin Islands.
Of those interviewed, 88% reported consuming moringa‑containing supplements.
Symptoms of Salmonella
Symptoms typically appear 12–72 hours after exposure and may include:
• Diarrhea
• Fever
• Abdominal cramps
Young children, older adults, and people with weakened immune systems face higher risk for severe illness. Anyone who develops symptoms after consuming recalled products should contact a healthcare provider.
- What Consumers and Retailers Should Do
- Consumers who have symptoms should contact their health care provider to report their symptoms and receive care.
Do not eat, sell, or serve recalled supplements.
Throw them away immediately.
Clean and sanitize any surfaces or containers the products touched.
Request refunds from the respective companies.
Retailers should remove recalled inventory and stop sales.
©2026 BCDollarSaver.com. All rights reserved.

Be the first to comment